Adipose tissue signaling research has become a major scientific focus because adipose depots function as active signaling systems rather than passive storage compartments. In controlled experimental models, researchers examine how adipocyte receptors, intracellular signaling cascades, lipid-handling pathways, transcriptional regulators, and adipokine networks coordinate the storage, mobilization, and redistribution of energy. These investigations often center on white adipose tissue, brown adipose tissue, and beige adipocyte biology, with particular attention to how receptor activation influences cAMP dynamics, kinase signaling, mitochondrial activity, adipokine secretion, and energy homeostasis modeling. Readers exploring this broader category can also review the Metabolic Research Hub for related pathway-focused content across metabolic peptide research.
In research settings, adipose tissue is studied as a highly responsive network that integrates hormonal, neural, nutrient, and inflammatory inputs. Rather than responding through a single linear mechanism, adipocytes coordinate multiple signaling systems at once, including adrenergic signaling, insulin-associated signaling, mTOR-linked nutrient sensing, lipolytic control systems, adipokine-mediated communication, and thermogenic transcription programs. This complexity is one reason adipose biology remains central to modern metabolic research. A broad review of adipose tissue biology and its role in energy homeostasis is available on PubMed in Advances in the understanding of adipose tissue biology, with additional literature discoverable through Google Scholar. :contentReference[oaicite:0]{index=0}
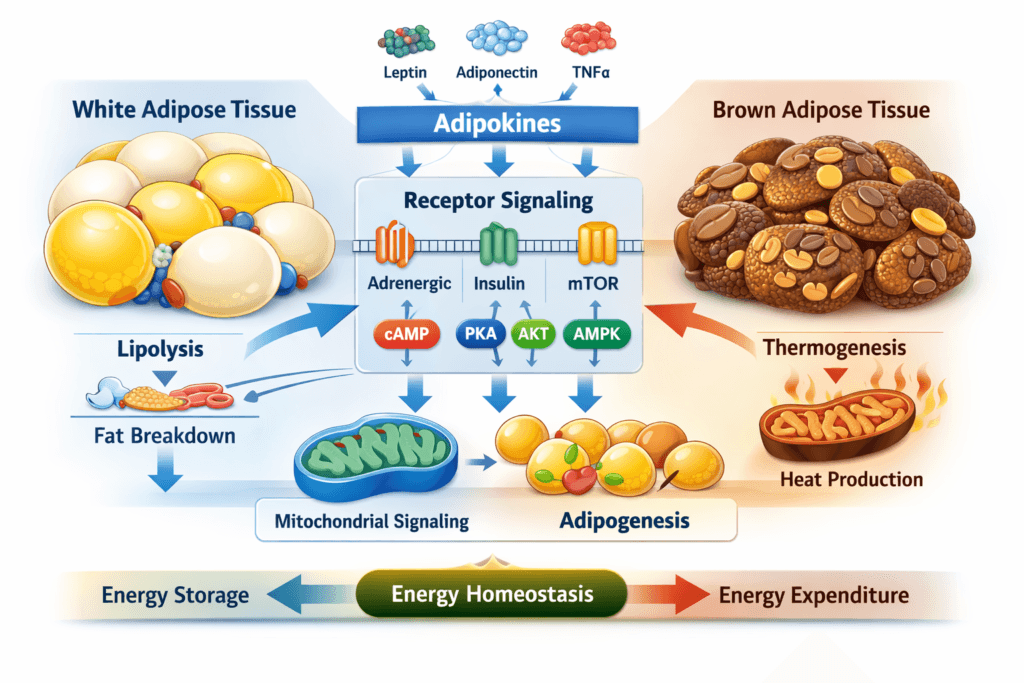
Adipose Tissue as a Signaling Organ
Modern adipose research consistently describes adipose tissue as an endocrine, paracrine, and metabolic signaling organ. Adipocytes secrete adipokines, lipokines, and other signaling mediators that influence communication with the brain, liver, skeletal muscle, pancreas, vascular tissues, and immune cell populations. This endocrine dimension has shifted adipose research away from older storage-only models and toward network-level interpretations of tissue function. A useful overview of adipose tissue as an endocrine signaling system is available in Adipose tissue: an endocrine organ playing a role in metabolic regulation, while a newer review addressing adipokine crosstalk is Adipokines in the Crosstalk between Adipose Tissues and Other Organs. Readers can also explore broader academic indexing through Google Scholar. :contentReference[oaicite:1]{index=1}
This signaling role is especially important in energy homeostasis modeling because adipose tissue does not simply receive instructions from other organs. It also sends information outward. Adipokine release, substrate flux, receptor-driven lipolysis, and thermogenic signaling can all reshape systemic metabolic context in preclinical experimental systems. That is why adipose signaling research often overlaps with endocrine physiology, mitochondrial biology, immune signaling, and nutrient-sensing pathways instead of being confined to a single narrow niche. For readers interested in related signaling architecture across metabolic pathways, the earlier article on GLP Receptor Signaling Mechanisms can help connect receptor pharmacology with broader metabolic network analysis.
Adipocyte Receptor Interactions and Signal Integration
Adipocyte receptor interactions are a major focus of preclinical research because adipocytes integrate input from multiple receptor systems at the plasma membrane and across intracellular compartments. Experimental studies often evaluate adrenergic receptors, insulin-linked pathways, natriuretic peptide signaling, adipokine receptors, cytokine-responsive pathways, and nutrient-sensing systems in order to understand how adipocytes choose between storage, release, remodeling, and thermogenic responses. Rather than acting independently, these receptor systems often converge on shared signaling nodes such as cAMP, PKA, PI3K-AKT, ERK, AMPK, and mTOR.
Reviews on adipocyte signaling repeatedly note that receptor context matters just as much as receptor presence. Signal strength, duration, cell state, depot type, and surrounding immune environment can all alter the final pathway readout. Gastrointestinal peptide signaling in adipose tissue has also been discussed in the literature as part of broader receptor-network investigation, as outlined in Gastrointestinal regulatory peptides and their effects on fat tissue. Related literature searches can be expanded through Google Scholar. :contentReference[oaicite:2]{index=2}
cAMP Signaling, Lipolysis, and Intracellular Adipocyte Dynamics
One of the most established intracellular themes in adipose tissue signaling research is cAMP-mediated control of lipolytic activity and pathway coordination. In adipocytes, cAMP is studied as a central second messenger that can redistribute signaling into multiple downstream effectors, particularly protein kinase A and scaffold-dependent regulatory systems. These pathways help control phosphorylation events associated with lipid droplet access, lipase coordination, perilipin dynamics, and broader substrate mobilization responses. Because adipose tissue serves as a major energy reservoir, cAMP-regulated lipolysis remains one of the most intensively studied features of adipocyte signaling.
Researchers also study cAMP compartmentalization in adipocytes because signal location can be as important as signal magnitude. A localized cAMP event near lipid droplets or mitochondrial interfaces may have very different consequences from a diffuse cytosolic rise. This concept is discussed in Compartmentalization of cAMP Signaling in Adipogenesis, Lipogenesis, and Lipolysis, while regulation of lipolytic restraint and pathway control is reviewed in The Molecular Brakes of Adipose Tissue Lipolysis. Additional papers can be explored through Google Scholar. :contentReference[oaicite:3]{index=3}
This intracellular perspective is important for energy homeostasis modeling because lipolysis is not merely a biochemical endpoint. It is part of a broader signaling decision regarding whether stored substrate remains in adipose tissue, is mobilized to other tissues, or is redirected into thermogenic processes. That is why adipocyte signaling research often links cAMP biology with mitochondrial function, lipid droplet remodeling, transcriptional adaptation, and inter-organ communication.
Adipogenesis, Differentiation, and Adipocyte Identity Programs
Adipose signaling research also includes intensive study of adipocyte differentiation and lineage programming. In preclinical systems, investigators track how precursor cells transition into mature adipocytes, how receptor inputs influence this progression, and how transcriptional networks establish white, brown, or beige cellular identity. These questions matter because adipose signaling capacity is strongly shaped by adipocyte state. Mature lipid-storing cells, thermogenic adipocytes, and differentiating preadipocytes can respond very differently to the same signaling cue.
Foundational reviews on adipocyte differentiation include Adipocyte differentiation: from fibroblast to endocrine cell, while broader work on adipocyte life cycle regulation is discussed in Phytochemicals and regulation of the adipocyte life cycle. The same themes can be extended through Google Scholar. These sources are useful because they frame adipogenesis as a signaling-regulated developmental process, not just a passive increase in lipid storage. :contentReference[oaicite:4]{index=4}
In practical research terms, this means adipose tissue modeling often includes both dynamic signaling assays and transcription-based identity studies. Researchers may evaluate receptor activation, kinase distribution, mitochondrial markers, and adipokine output alongside differentiation state in order to build a fuller picture of adipose function across experimental conditions.
Adipokines, Lipokines, and Inter-Organ Crosstalk
Adipokine and lipokine signaling remains one of the defining features of adipose tissue biology. Adipose-derived signaling molecules participate in communication with skeletal muscle, liver, vasculature, brain, gut, pancreas, and immune compartments. This inter-organ crosstalk is central to how adipose tissue contributes to energy homeostasis in research models. Rather than being isolated within adipose depots, signaling events in adipocytes can influence whole-body substrate handling, inflammatory tone, and endocrine communication across multiple tissues.
A recent review summarizing adipose tissue adipokines and lipokines is Adipose tissue adipokines and lipokines: Functions and crosstalk. Earlier work also established the importance of adipokines for energy homeostasis and metabolic regulation in Adipokines: Therapeutic Targets for Metabolic Syndrome. Readers wanting a broader literature map can review Google Scholar. Together, these sources support the modern view that adipose signaling research must account for both local tissue behavior and systemic communication. :contentReference[oaicite:5]{index=5}
This is one reason adipose signaling pages fit naturally inside a larger metabolic research cluster. A reader who begins with receptor signaling, adipokine biology, or adipocyte pathway modeling may also benefit from browsing category-specific resources within the Metabolic Research Hub, where signaling-oriented content can be connected to broader metabolic research materials.
Brown Adipose Tissue, Thermogenesis, and Energy Dissipation
Brown adipose tissue research adds another major dimension to adipose signaling biology because it shifts the discussion from energy storage toward energy dissipation. Brown adipocytes are studied for their thermogenic capacity, mitochondrial density, and responsiveness to sympathetic signaling. This makes brown adipose tissue especially important in energy homeostasis modeling, where investigators want to understand how signaling pathways regulate substrate use, heat generation, and depot-specific specialization. A classic foundational review is Brown adipose tissue: function and physiological significance, with additional pathway-focused coverage in Signaling Pathways Regulating Thermogenesis. Broader discovery results are also available through Google Scholar. :contentReference[oaicite:6]{index=6}
More recent research continues to expand this picture by examining adrenergic signaling, mitochondrial remodeling, futile substrate cycles, and signaling pathways that regulate thermogenic competence over time. A 2025 review on brown adipose adrenergic regulation is available in PubMed at mTORC1 and 2 Adrenergic Regulation and Function in Brown Adipose Tissue, while another review covering thermogenic differentiation is Non-shivering Thermogenesis Signalling Regulation. These studies help show why adipose tissue research increasingly separates depot-specific signaling questions instead of treating all adipose tissue as functionally identical. :contentReference[oaicite:7]{index=7}
mTOR, Nutrient Sensing, and Adipose Pathway Coordination
Nutrient-sensing pathways are also central to adipose tissue signaling research, with mTOR signaling serving as one of the most important examples. mTOR-associated pathways are studied because they connect nutrient availability, growth-state signaling, adipocyte differentiation, and metabolic adaptation. In adipose tissue, altered mTOR behavior can influence tissue expansion, lipid handling, and broader homeostatic pathway coordination. The review Recent Advances in Adipose mTOR Signaling and Function provides a useful summary, and additional literature can be browsed via Google Scholar. :contentReference[oaicite:8]{index=8}
Researchers often evaluate mTOR together with adrenergic inputs, insulin-linked signals, lipolytic regulators, and mitochondrial pathways because adipose tissue rarely responds through one pathway in isolation. This systems view supports more realistic energy homeostasis models and helps explain why adipose signaling research continues to expand into multi-pathway integration rather than single-receptor analysis alone.
Immune Signaling and Adipose Microenvironment Research
Another important development in this field is the recognition that adipose signaling is strongly shaped by the local microenvironment, including resident immune cells. Reviews on immunologic and endocrine functions of adipose tissue describe how shifts in adipocyte state can influence neighboring immune populations and vice versa, altering local signaling tone and broader tissue behavior. A useful overview is Immunologic and endocrine functions of adipose tissue. More specific work on immune-cell signaling in thermogenic adipose biology is discussed in Immune cell cholinergic signaling in adipose thermoregulation, with related search results on Google Scholar. :contentReference[oaicite:9]{index=9}
This immune-adipose interface is especially valuable for preclinical modeling because it makes clear that adipose signaling cannot be understood only by measuring isolated adipocytes. Tissue context, inflammatory tone, and cell-cell communication all shape how receptor activation is translated into measurable pathway outcomes.
Why Adipose Tissue Signaling Matters in Metabolic Research
Adipose tissue signaling matters because adipose depots sit at the intersection of substrate storage, energy release, inter-organ communication, and thermogenic adaptation. For that reason, adipose research connects naturally with the broader metabolic research category. Readers working through these topics may also explore category-related Synagenics metabolic pages such as GLP-3RT, GLP-2TZ, Cargrilintide 5mg, Survodutide, AOD-9604, 5-Amino-1MQ 5mg, Tesamorelin 10mg, LipoBlaster, and Lipo-CB12.
These product links should sit within a larger educational structure rather than stand alone. Internal authority improves when mechanism pages, overview articles, and research-category hubs support each other through clean contextual linking. For that reason, this page pairs well with the Metabolic Research Hub, the GLP-3RT Research Hub, and practical workflow resources like the Reconstitution Calculator.
Conclusion
Adipose tissue signaling research continues to expand because adipose biology is now understood as a deeply integrated signaling system rather than a passive metabolic reserve. In preclinical experimental models, adipocyte receptor interactions, cAMP-regulated lipolysis, nutrient-sensing pathways, adipokine release, depot-specific thermogenic programs, and immune microenvironment cues all contribute to the final tissue response. This is why adipose pathway analysis remains so important for energy homeostasis modeling and broader metabolic research interpretation.
As the literature grows, the field continues moving toward more complete models that account for receptor context, intracellular pathway distribution, tissue specialization, and inter-organ communication. That broader perspective helps researchers understand how white, brown, and beige adipose systems behave under different experimental conditions and why adipose signaling remains one of the most important mechanistic themes in modern metabolic science. For broader cluster navigation, readers can continue through the Metabolic Research Hub and related pathway resources referenced throughout this page.
Selected Sources and Further Reading
- Advances in the understanding of adipose tissue biology — PubMed | Google Scholar
- Adipose tissue: an endocrine organ playing a role in metabolic regulation — PubMed | Google Scholar
- Adipokines in the Crosstalk between Adipose Tissues and Other Organs — PubMed | Google Scholar
- Compartmentalization of cAMP Signaling in Adipogenesis, Lipogenesis, and Lipolysis — PubMed | Google Scholar
- The Molecular Brakes of Adipose Tissue Lipolysis — PubMed | Google Scholar
- Brown adipose tissue: function and physiological significance — PubMed | Google Scholar
- Signaling Pathways Regulating Thermogenesis — PubMed | Google Scholar
- Recent Advances in Adipose mTOR Signaling and Function — PubMed | Google Scholar
Research Use Notice: This content is presented for educational and informational purposes within a research context. Products referenced on Synagenics are intended for research use only.
