GLP receptor signaling mechanisms are a major focus in peptide research because they connect receptor pharmacology, second-messenger generation, intracellular trafficking, and pathway-specific downstream responses into one highly coordinated signaling framework. In controlled research models, investigators examine how GLP-1 receptor, GIP receptor, and glucagon receptor activation influences cyclic AMP production, protein kinase activation, β-arrestin recruitment, ERK signaling, endosomal activity, receptor recycling, and transcription-associated cascade behavior. Rather than functioning as simple on-off switches, these receptors behave as dynamic signaling platforms whose output can shift according to ligand structure, receptor conformation, cellular background, membrane localization, and duration of exposure. Readers exploring the broader category can also review the Metabolic Research Hub for additional pathway-based resources and related content.
This area of research has become especially important because GLP-1, GIP, and glucagon receptor systems overlap at several signaling nodes while still maintaining distinct intracellular identities. Shared engagement of Gαs and cAMP does not mean identical downstream behavior. Researchers continue to document important differences in signaling amplitude, signal persistence, β-arrestin dependency, receptor internalization, ERK activation, and compartmentalized cAMP activity across this receptor family. Those distinctions help explain why single agonists, dual agonists, and multi-receptor research compounds can generate different intracellular signatures even when they belong to the same broader metabolic signaling landscape. For additional internal context, readers may also review the GLP-3RT Research Hub and the Reconstitution Calculator.
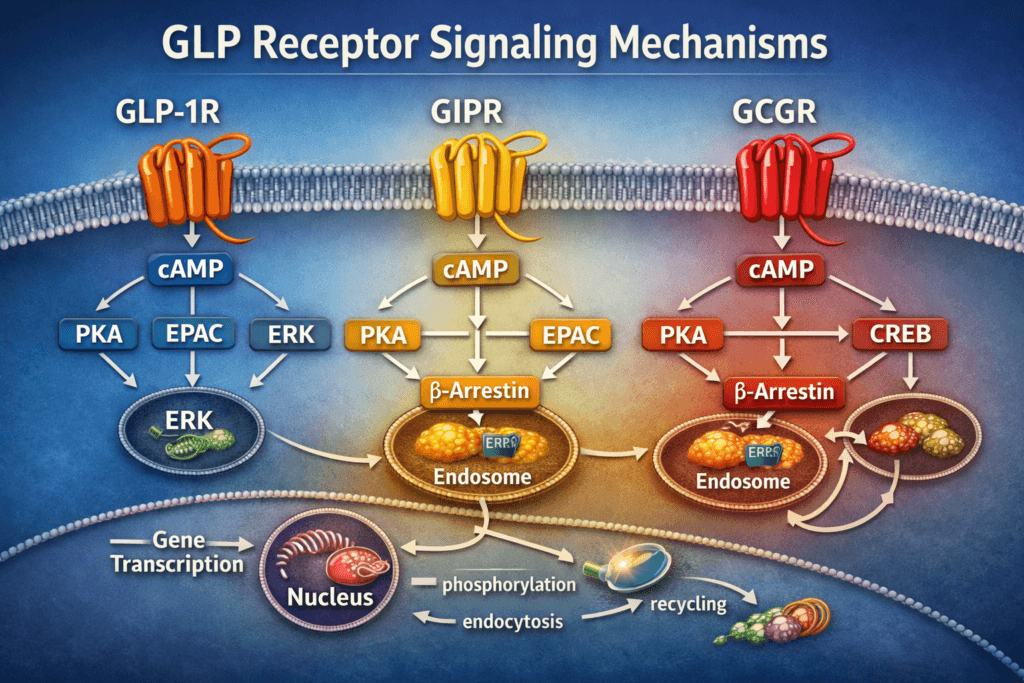
Class B GPCR Structure and Receptor Family Context
GLP-1 receptor, GIP receptor, and glucagon receptor belong to the class B family of G protein-coupled receptors, a receptor group known for peptide-sensitive extracellular domains and complex intracellular signaling behavior. These receptors are often studied together because they share structural similarities and because each can initiate strong cAMP-linked signaling. At the same time, modern research continues to show that receptor family similarity does not eliminate signaling diversity. Distinct ligand-binding modes, conformational shifts, receptor trafficking patterns, and scaffold interactions can all alter how signaling unfolds after activation. A strong review on GLP-1 receptor cAMP biology and signaling compartmentalization is available on PubMed here: New Insights into Beta-Cell GLP-1 Receptor and cAMP Signaling. A related literature search can also be explored through Google Scholar. :contentReference[oaicite:0]{index=0}
Comparative signaling studies are particularly useful because they show that shared incretin-associated receptors can still diverge in arrestin recruitment, trafficking kinetics, and downstream pathway emphasis. That comparative framework is one reason the field increasingly evaluates receptor signaling through multi-parameter assays instead of relying only on a single cAMP endpoint. Research reviews focusing on both GLP-1R and GIPR support this broader interpretation and help explain why co-activation systems are now studied as integrated signaling networks rather than isolated receptor events. See GLP-1 and GIP receptor signaling in beta cells on PubMed and the corresponding Google Scholar search. :contentReference[oaicite:1]{index=1}
GLP-1 Receptor Activation and cAMP Signaling
The best-characterized signaling feature of GLP-1 receptor activation is coupling to Gαs, which stimulates adenylyl cyclase and increases intracellular cAMP. In research settings, cAMP is treated as a central organizing second messenger rather than a simple isolated readout. Investigators often examine not only how much cAMP is generated, but also where it accumulates inside the cell, how long it persists, and which downstream effectors it preferentially engages. This matters because two ligands may both increase cAMP while still producing very different trafficking and signaling outcomes. The importance of cAMP compartmentalization, receptor internalization, and signal bias is discussed in this PubMed review, while broader academic search results can be browsed through Google Scholar. :contentReference[oaicite:2]{index=2}
Once cAMP is generated, it can activate multiple intracellular arms, with protein kinase A and exchange protein directly activated by cAMP, or EPAC, being two of the best known. PKA is associated with phosphorylation-based signal relay and amplification, while EPAC supports additional cAMP-responsive functions that may unfold with different timing or spatial patterning. This distinction remains central to GLP receptor signaling research because it helps explain why cAMP elevation alone does not fully predict intracellular behavior. A classic PubMed source addressing this framework is Epac: A new cAMP-binding protein in support of glucagon-like peptide-1 receptor research, with a matching Google Scholar search here. :contentReference[oaicite:3]{index=3}
GIP Receptor Signaling and Comparative Incretin Pharmacology
GIP receptor signaling is frequently examined alongside GLP-1 receptor signaling because both receptors participate in incretin-associated research and both strongly engage cAMP pathways through Gαs. However, side-by-side analysis increasingly shows that the two receptors are not intracellular duplicates. Even where there is overlap in second-messenger generation, there may still be meaningful divergence in trafficking behavior, β-arrestin involvement, receptor recycling, and downstream pathway preference. This is highly relevant in studies involving co-agonists and dual-pathway constructs because receptor interaction cannot be reduced to a simple additive model. PubMed coverage of this topic is available in GLP-1 and GIP receptor signaling in beta cells, with additional discovery results through Google Scholar. :contentReference[oaicite:4]{index=4}
More recent work has further clarified that GLP-1R and GIPR can signal through distinct β-arrestin-dependent pathways. That is important because β-arrestins do more than terminate G protein signaling. They can also organize receptor trafficking and scaffold downstream pathways, influencing how a signal is distributed across time and cellular space. When researchers compare these receptors under the same experimental conditions, the differences that appear can help explain why multi-receptor research constructs produce distinct assay profiles. A strong paper for this comparison is GLP-1 and GIP receptors signal through distinct β-arrestin 2-dependent pathways to regulate pancreatic β cell function, with supporting search results available through Google Scholar. :contentReference[oaicite:5]{index=5}
Glucagon Receptor Signaling and Transcription-Linked Cascade Behavior
Glucagon receptor signaling contributes another major arm to this receptor family network. Like GLP-1R and GIPR, the glucagon receptor can drive cAMP production through Gαs. However, research often highlights its strong relationship to transcription-associated signaling, especially the cAMP-PKA-CREB axis. For this reason, glucagon receptor studies are highly relevant when researchers want to understand how acute receptor activation translates into longer-range intracellular adaptation and gene-regulatory output. A useful review is The Three Ds of Transcription Activation by Glucagon, and broader literature can be explored through Google Scholar. :contentReference[oaicite:6]{index=6}
Additional experimental work also shows that glucagon can engage a PKA/ERK signaling network linked to CREB phosphorylation under certain conditions. This makes glucagon receptor signaling especially useful in studies looking at receptor cross-network behavior, pathway bias, and temporal sequence of downstream activation. Rather than existing as a parallel version of GLP-1R signaling, glucagon receptor signaling adds a distinct layer of transcription-linked and kinase-integrated output to the broader metabolic receptor system. A relevant PubMed article is Glucagon promotes cAMP-response element-binding protein phosphorylation via a PKA/ERK1/2 signaling pathway, with a matching Google Scholar search. :contentReference[oaicite:7]{index=7}
ERK, Kinase Relays, and Signal Distribution Beyond cAMP
One of the most important principles in receptor pharmacology is that cAMP is not the end of the pathway. Instead, it serves as a central node that can distribute signaling into additional cascades, including kinase-linked pathways such as ERK1/2. In GLP receptor research, ERK activation has been especially important because it can arise through more than one intracellular route. Some studies describe PKA-associated ERK activation, while others support a role for β-arrestin-scaffolded signaling. These distinct routes may operate with different timing and localization, reinforcing the idea that receptor signaling should be understood as a layered process rather than a single linear sequence. Broader summaries of signaling network variation are discussed in Specific actions of GLP-1 receptor agonists and DPP4 inhibitors to regulate pancreatic β cell mass and function, and more literature can be reviewed through Google Scholar. :contentReference[oaicite:8]{index=8}
This multi-branch behavior is one reason researchers increasingly compare ligands using more than receptor binding and cAMP accumulation. They also examine ERK phosphorylation, arrestin recruitment, internalization kinetics, and recovery dynamics. By doing so, they can identify signaling differences that would be invisible in a simple potency screen. This richer approach is especially valuable when analyzing receptor-selective ligands versus dual- or tri-receptor constructs within metabolic peptide research.
β-Arrestin Recruitment, Internalization, and Endosomal Signaling
β-Arrestin biology has become one of the most important topics in GLP receptor signaling research. β-Arrestins were once discussed mainly as desensitization adaptors that uncouple receptors from G proteins and support internalization, but current literature shows a more complex role. They can also scaffold downstream signaling and shape whether a receptor continues to signal after internalization. In GLP-1R systems, β-arrestins influence receptor trafficking, recycling, intracellular signaling persistence, and the balance between transient surface signaling and sustained endosomal activity. Studies in PubMed and broader academic literature continue to support this expanded view of receptor regulation. See Abolishing β-arrestin recruitment is necessary for the full efficacy of a G protein-biased GLP-1R agonist and the related Google Scholar search. :contentReference[oaicite:9]{index=9}
Endosomal signaling is another major development in this area. Research now supports the idea that certain GPCRs, including GLP-1R, can continue signaling after internalization instead of becoming immediately silent once removed from the plasma membrane. This changes how investigators interpret signal duration and receptor efficacy because internalized receptors may still generate intracellular information. A relevant study on disruption of GLP-1R internalization and endosomal signaling is Engineered mini-G proteins block the internalization of GLP-1R and associated intracellular signaling, with more literature accessible through Google Scholar. :contentReference[oaicite:10]{index=10}
Why These Mechanisms Matter in Multi-Receptor Metabolic Research
The convergence of GLP-1R, GIPR, and GCGR research has shifted the field toward network-level interpretation. Investigators no longer assume that strong activity at one receptor is enough to describe an entire signaling profile. Instead, they examine receptor balance, temporal coordination, pathway bias, trafficking differences, and transcription-associated output. This is especially relevant when evaluating compounds associated with metabolic peptide research, where multi-receptor design and overlapping signaling themes are common. Readers exploring category-relevant materials can review Synagenics metabolic products such as GLP-3RT, GLP-2TZ, Cargrilintide 5mg, Survodutide, AOD-9604, 5-Amino-1MQ 5mg, and Tesamorelin 10mg.
Additional metabolic category pages may also be helpful for readers organizing their research workflow, including LipoBlaster and Lipo-CB12. Category-level structure remains important because it helps readers move from broad signaling concepts into narrower product and pathway discussions. For that reason, linking this article back to the Metabolic Research Hub strengthens the overall research cluster. If the previous article you want linked is your earlier metabolic overview page, replace the placeholder below with that live slug once published: Metabolic Peptide Research Overview.
Conclusion
GLP receptor signaling mechanisms remain central to metabolic peptide research because they show how receptor activation is translated into layered intracellular behavior. GLP-1 receptor, GIP receptor, and glucagon receptor pathways all interact with cAMP-linked signaling, yet each can produce distinct profiles of kinase engagement, β-arrestin recruitment, internalization, endosomal signaling, and transcription-associated output. These differences are exactly why detailed receptor pharmacology remains so valuable in controlled laboratory settings.
As the field develops, research continues to move beyond simple potency-based interpretation and toward models that account for spatial signaling, pathway bias, receptor trafficking, and network integration. That broader perspective makes it easier to understand why related receptors may behave differently, why multi-receptor constructs require deeper mechanistic analysis, and why signaling architecture matters as much as receptor binding itself. For readers building a broader understanding of this category, the best approach is to combine primary literature, comparative signaling studies, and structured internal resources such as the Metabolic Research Hub, the GLP-3RT Research Hub, and the Reconstitution Calculator. :contentReference[oaicite:11]{index=11}
Selected Sources and Further Reading
- New Insights into Beta-Cell GLP-1 Receptor and cAMP Signaling — PubMed | Google Scholar
- GLP-1 and GIP receptor signaling in beta cells — PubMed | Google Scholar
- Epac: A new cAMP-binding protein in support of glucagon-like peptide-1 receptor research — PubMed | Google Scholar
- GLP-1 and GIP receptors signal through distinct β-arrestin 2-dependent pathways — PubMed | Google Scholar
- The Three Ds of Transcription Activation by Glucagon — PubMed | Google Scholar
- Glucagon promotes CREB phosphorylation via PKA/ERK1/2 signaling — PubMed | Google Scholar
- Abolishing β-arrestin recruitment is necessary for the full efficacy of a G protein-biased GLP-1R agonist — PubMed | Google Scholar
- Engineered mini-G proteins block GLP-1R internalization and intracellular signaling — PubMed | Google Scholar
Research Use Notice: This content is provided for educational and informational purposes within a research context. Products referenced on Synagenics are intended for research use only.
